Aleafia Health Study Indicates 45% of Patients End Benzodiazepine Use Following Medical Cannabis Treatment
- First of its Kind Study Published in Peer-Reviewed Journal, “Cannabis and Cannabinoid Research”
- Authors Leveraged Aleafia’s 10 Million Point Medical Cannabis Dataset
TORONTO, January 25, 2019 – Aleafia Health Inc. (TSXV: ALEF, OTC: ALEAF, FRA: ARAH) (“Aleafia Health” or the “Company”), announces today what it believes to be the first study reporting reduced benzodiazepine use among patients initiated and monitored on medical cannabis. Benzodiazepines, a class of psychoactive drugs, include the most common sedatives and anti-anxiety medications.
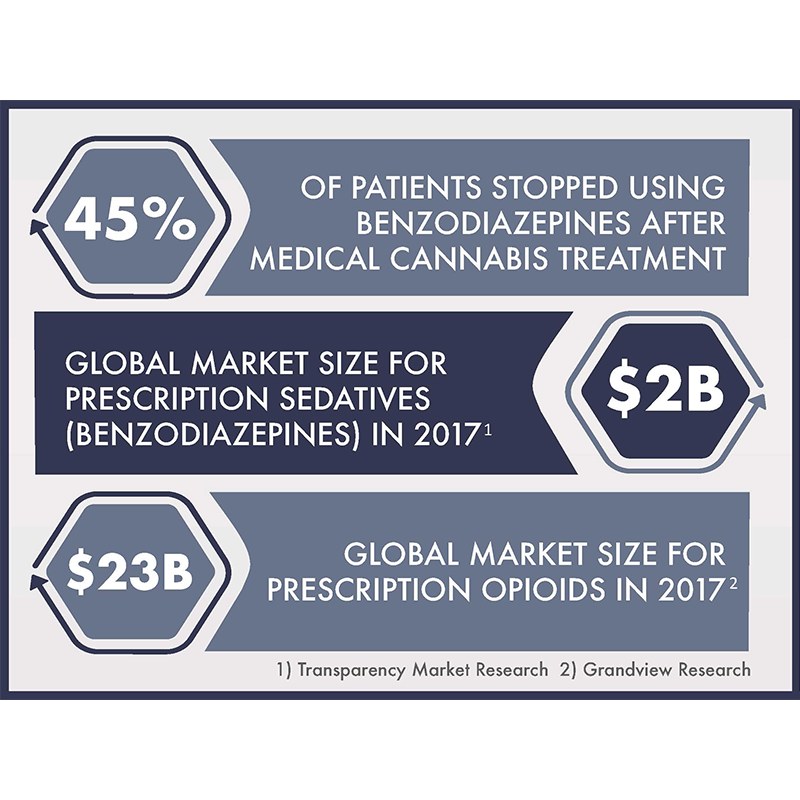
The retrospective study, published in Cannabis and Cannabinoid Research, a peer-reviewed journal, is entitled “Reduction of Benzodiazepine Use in Patients Prescribed Medical Cannabis.” It found that 45.2 per cent of patients regularly consuming benzodiazepines had stopped taking the medication within approximately six months of beginning medical cannabis. These patients, following prescription cannabis use, also reported decreased daily distress due to medical conditions.
The cohort featured 146 patients who received physician-led treatment in Aleafia Health’s wholly owned Canabo Medical Clinics (“Canabo”). Aleafia believes it maintains the world’s largest medical cannabis patient dataset and is deploying the data for unique insights in medical cannabis treatment and product development best practices.
The Canadian Centre on Substance Abuse and Addiction (CCSAA) warns that benzodiazepines may lead to serious long-term complications, including dependence, overdose and death. According to a CCSAA study, 10 per cent of the Canadian population takes prescription sedatives. And a study published in The British Medical Journal found that Benzodiazepines consumption significantly increased the risk of opioid overdose.
“The study results are encouraging, and this work is concurrent with growing public interest in a rapidly developing Canadian cannabis market,” said lead author Chad Purcell. “We are advising the public to observe caution. The results do not suggest that cannabis should be used an alternative to conventional therapies. Our purpose is inspiring others to advance current cannabis understanding as we collect stronger efficacy and safety data that will lead to responsible policy and recommended practices for use.”
“Due to regulatory hurdles and lingering stigma, there remains a shortage of medical cannabis research. At Aleafia, we will continue to leverage our IP and leading cannabis data to further advance patient care through advanced treatment methods and specialized product development,” said Aleafia Health Chief Medical Officer Dr. Michael Verbora. “This study’s results will not be surprising to many patients who have transitioned from prescription pain killers and sedatives with the help of physician-led medical cannabis therapy. I thank the study authors for this significant contribution to evidence-based medicine.”
For Investor & Media Relations, please contact:
Nicholas Bergamini, VP, Public Affairs
416-860-5665
About the Authors:
Chad Purcell BSc (Pharm)
Chad Purcell will receive an MD degree from Dalhousie University in 2019 from which he also earned a BSc with honours in pharmacology and, subsequently, a BSc in pharmacy. He has published in several areas of pharmaceutical research including pharmacokinetics, pharmacy practice and most recently, medical cannabis.
Dr. Andrew Davis
Dr. Andrew Davis received a Ph.D. and M.A. in Economics from the University of Rochester, where he also held both a Sproull and Lionel McKenzie Fellowships. He also earned a B.Sc. from Memorial University of Newfoundland. Dr. Davis has regularly presented research at a variety of seminars and conferences including the Canadian Economics Association, of which he is a member, and is most recently published in Economic Modelling.
Dr. Nico Moolman, MD
Dr. Nico Moolman is a Clinical Assistant Professor at the University of Saskatchewan in Saskatoon. He specializes in Otolaryngology – Head and Neck surgery with a focus on Sinonasal, as well as Facial Plastic and Reconstructive surgery. His research has focused on Head and Neck Oncology with publications in the field.
Dr. Mark Taylor, MD
Dr. Mark Taylor, currently a Professor as well as the Interim Head of the Division of Otolaryngology-Head and Neck Surgery at Dalhousie University, has 15 years’ experience as a Head and Neck Surgeon in Halifax. In addition to resident education, his practice focuses on Head and Neck Oncology, as well as Facial Plastic and Reconstructive Surgery, in which he is also Board Certified. Dr. Taylor has over 150 peer- reviewed publications to his credit and has developed a particular interest in research into cannabinoid therapy.
About Aleafia Health:
Aleafia, a vertically integrated, national cannabis company, operates major medical clinic, cannabis cultivation and R&D facilities. A federally licensed producer and vendor of cannabis, the company has a planned and fully-funded annual production capacity of 98,000 kg of dried cannabis flower.
Aleafia operates medical cannabis clinics staffed by physicians and nurse practitioners, with over 50,000 patients. The company is highly differentiated, maintaining the largest medical cannabis dataset in the world.
Neither the TSX Venture Exchange nor its Regulation Services Provider (as that term is defined in the policies of the Exchange) accepts responsibility for the adequacy or accuracy of this release.
FORWARD LOOKING INFORMATION
This press release contains forward-looking statements and information that are based on the beliefs of management and reflect the Company’s current expectations. When used in this press release, the words “estimate”, “project”, “belief”, “anticipate”, “intend”, “expect”, “plan”, “predict”, “may” or “should” and the negative of these words or such variations thereon or comparable terminology are intended to identify forward-looking statements and information. The forward-looking statements and information in this press release includes information relating to the implementation of Aleafia Health’s business plan. Such statements and information reflect the current view of the Company with respect to risks and uncertainties that may cause actual results to differ materially from those contemplated in those forward-looking statements and information.
By their nature, forward-looking statements involve known and unknown risks, uncertainties and other factors which may cause our actual results, performance or achievements, or other future events, to be materially different from any future results, performance or achievements expressed or implied by such forward-looking statements. Such factors include, among others, the following risks: risks associated with the implementation of Aleafia Health’s business plan and matters relating thereto, risks associated with the cannabis industry, competition, regulatory change, the need for additional financing, reliance on key personnel, the potential for conflicts of interest among certain officers or directors, and the volatility of the Company’s common share price and volume. Forward-looking statements are made based on management’s beliefs, estimates and opinions on the date that statements are made, and the Company undertakes no obligation to update forward-looking statements if these beliefs, estimates and opinions or other circumstances should change. Investors are cautioned against attributing undue certainty to forward-looking statements.
